
What is an atom?Īt the very center of an atom is the nucleus, which is made up of small particles called protons and neutrons. 5A: Solubility and Solution Reactions (Worksheet) So far, we have considered stoichiometric relationships on the basis of masses and moles.Atoms are the building blocks of the universe they make up everything you see around you.Understanding the mechanism allows chemists to devise ways of optimizing chemical reactions. This knowledge can be used to gain insight into the detailed molecular pathway (the mechanism) by which the reaction occurs. Experimentally determined rate law expressions show how the rate of a reaction depends upon the concentrations of the reactants and sometimes the products, too. 4B: Kinetics I (Worksheet) Chemical kinetics is the study of the rates of chemical reactions.Next to the atomic theory, the mole concept is the most fundamental unifying idea in all chemistry. To deal with the very large numbers of atoms and molecules in such samples, chemists developed the unit of the mole (abbreviated mol) and a unit of measure called the molar mass, which has units of g/mol. 4A: Moles & Stoichiometry (Worksheet) Chemists are concerned with mass relationships in chemical reactions, usually run on a macroscopic scale (grams, kilograms, etc.).The strength of these attractions also determines what changes in temperature and pressure are needed to effect a phase transition. The phase that we see under ordinary conditions (room temperature and normal atmospheric pressure) is a result of the forces of attraction between molecules or ions comprising the substance. 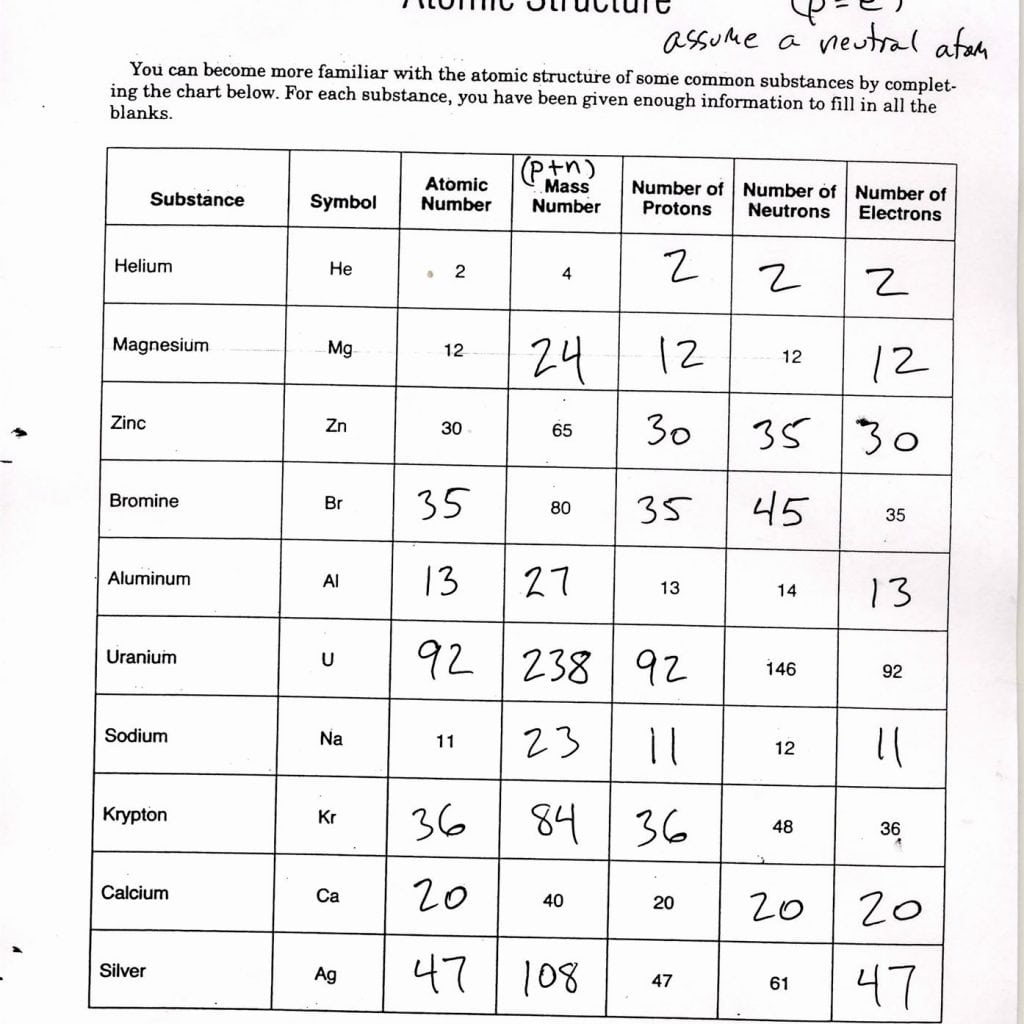
3B: Intermolecular Forces - Liquids, Solids, and Solutions (Worksheet) Most substances can exist in either gas, liquid, or solid phase under appropriate conditions of temperature and pressure.Knowing these fundamental molecular or ionic unit masses allows us to predict mass changes that occur on the macroscopic scale as a result of chemical reactions. Describing the fundamental compound unit as either a molecule or a formula unit allows us to determine the mass of that unit. Knowing the classification allows us to name the compound correctly and to understand the microscopic organization of it. 3A: Compounds, Naming, Reaction Equations, and Formula Weights (Worksheet) Compounds are generally classified as molecular, ionic, or (more rarely) network.This and all of the behaviors represented by PV = nRT can be understood on the basis of a model called the Kinetic Molecular Theory. 2B: Gas Laws II (Worksheet) The fundamental relationship PV = nRT can be extended to understand the densities of gases under various conditions and to understand how non-reacting gases behave when mixed together.To understand how atoms combine to form compounds, you need to understand their basic composition and structure. The mass relationships between elements and compounds in chemical reactions ultimately relate back to the characteristics of the atoms of which they are composed. Atoms are the fundamental building blocks of all matter. 2A: Basic Atomic Structure (Worksheet) The atomic theory of matter is the great organizing principle of chemistry.This shows the relationship between a gas’s pressure (P), temperature (T), volume (V), and amount in moles (n). The observed behavior of gases, embodied in the empirical gas laws, leads to a series of equations that can be summarized by a single equation of state, called the ideal gas law equation. 1B: Gas Laws - Part 1 (Worksheet) Of the three principal states of matter (gas, liquid, solid), gases show behavior that is most easily connected to molecular motion.But regardless of the units used, we want to have some confidence that our measured and calculated results bear a close relationship to the “true” values. These units facilitate international communication by discouraging use of units peculiar to one culture or another (e.g., pounds, inches, degrees Fahrenheit). Since 1960, the metric system in use has been the Système International d'Unités, commonly called the SI units. 1A: Units, Measurement Uncertainty, and Significant Figures (Worksheet) All scientists the world over use metric units.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
